Author: Lydia Penhaligon - Page 4
-
17 Dec

-
16 Dec

Why Prices Drop at Launch: The Real Reason Behind First Generic Entry
First generic entry causes prices to drop sharply at launch because competitors offer similar products at a fraction of the cost. This trend is reshaping software, electronics, and even pharmaceuticals. -
12 Dec
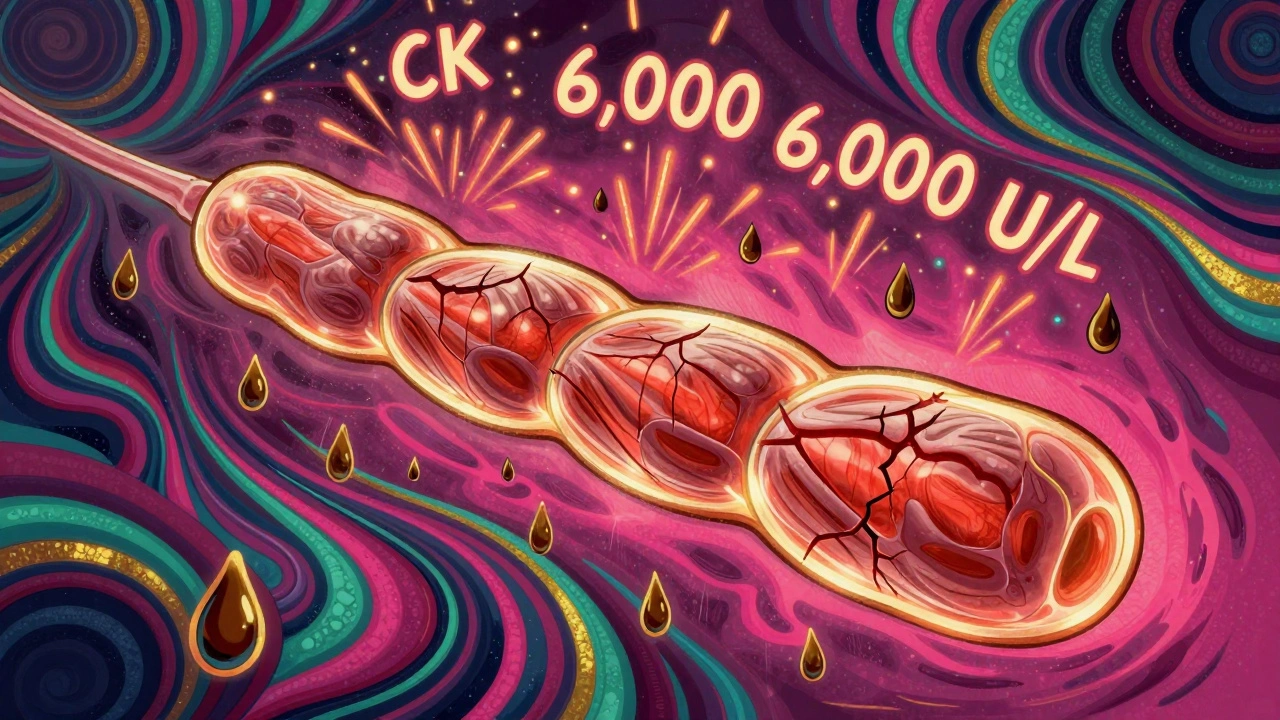
Daptomycin Muscle Toxicity: What You Need to Know About CK Monitoring and Symptoms
Daptomycin is effective against resistant infections but can cause muscle toxicity. Learn key symptoms, when to check CK levels, how to prevent serious damage, and what to do if you're on statins or higher doses. -
11 Dec

Medication Therapy Management Services Explained for Patients
Medication Therapy Management (MTM) is a free service for Medicare Part D patients taking multiple medications. Learn how pharmacists help you avoid side effects, save money, and take your drugs safely and correctly. -
9 Dec
Benzodiazepines: Memory Loss, Fall Risks, and How to Taper Safely
Benzodiazepines can cause memory loss, increase fall risk in older adults, and lead to dependence. Learn how tapering slowly can help restore cognitive function and reduce danger. -
8 Dec

Rhodiola and Antidepressants: What You Need to Know About Serotonin Risks
Rhodiola may help with mild depression, but combining it with antidepressants like SSRIs can cause serotonin syndrome-a dangerous, potentially fatal reaction. Learn the risks, symptoms, and why most supplements don't warn you. -
7 Dec
GLP-1 Agonists and Gallbladder Disease: Recognizing Abdominal Pain Red Flags
GLP-1 agonists like Ozempic and Wegovy can increase gallbladder disease risk, especially with rapid weight loss. Learn the key abdominal pain signs that signal gallstones or cholecystitis - and what to do next. -
5 Dec

Serious vs Non-Serious Adverse Events: When to Report in Clinical Trials
Understand when to report serious vs non-serious adverse events in clinical trials. Learn the FDA and ICH criteria, reporting timelines, common mistakes, and how to avoid costly errors in safety monitoring. -
4 Dec

Dry Eyes from Medications: Lubrication and Lifestyle Tips to Relieve Symptoms
Medications like antihistamines, antidepressants, and blood pressure pills can cause dry eyes. Learn how preservative-free tears, warm compresses, omega-3s, and lifestyle changes can relieve symptoms without stopping your prescriptions. -
4 Dec

Paragraph IV Certifications: How Generic Drug Makers Challenge Patents Before Launch
Paragraph IV certifications let generic drug makers challenge brand patents before launch, speeding up affordable drug access. This legal tool under the Hatch-Waxman Act saves billions annually but faces challenges from patent thickets and pay-for-delay deals. -
4 Dec

Stability Testing: Long-Term Quality Monitoring Post-Manufacture in Pharmaceuticals
Stability testing ensures pharmaceutical products remain safe and effective over time. Learn how real-time monitoring, ICH guidelines, and lab data determine shelf life and prevent dangerous drug failures. -
4 Dec

Postpartum Anxiety: Recognizing Symptoms, Screening Tools, and Effective Care Paths
Postpartum anxiety affects 1 in 5 new mothers and is often missed. Learn the key symptoms, how screening works, and proven care paths-from therapy to medication-that actually help.